
Question 3.9:The molar conductivity of 0.025 mol L−1 methanoic acid is 46.1 S cm2 mol−1. Calculate its degree of dissociation and dissociation constant. Given λ0(H+)= 349.6 S cm2 mol–1 and λ0(HCOO–) = 54.6 S cm2 mol–1 Chapter 3: Electrochemistry Chemistry Class 12 solutions are developed for assisting understudies with working on their score and increase knowledge of the subjects. The molar conductivity of 0.025 mol L?1 methanoic acid is 46.1 S cm2 mol?1. Calculate its degree of dissociation and dissociation constant. Given ?0(H+)= 349.6 S cm2 mol?1 and ?0(HCOO?) = 54.6 S cm2 mol?1 is solved by our expert teachers. You can get ncert solutions and notes for class 12 chapter 3 absolutely free. NCERT Solutions for class 12 Chemistry Chapter 3: Electrochemistry is very essencial for getting good marks in CBSE Board examinations
Question 3.9:The molar conductivity of 0.025 mol L−1 methanoic acid is 46.1 S cm2 mol−1. Calculate its degree of dissociation and dissociation constant. Given λ0(H+)= 349.6 S cm2 mol–1 and λ0(HCOO–) = 54.6 S cm2 mol–1
Answer:Given that
λ0(H+)= 349.6 S cm2 mol–1
λ0(HCOO–) = 54.6 S cm2 mol–1
Concentration ,C = 0.025 mol L-1
λ(HCOOH) = 46.1 S cm2 mol−1
use formula
λ°(HCOOH) = λ0(H+) + λ0(HCOO–)
plug the values we get
λ°(HCOOH) = 0.349.6 + 54.6
=404.2 S cm2 mol−1
Formula of degree of dissociation:
ά = λ°(HCOOH)/ λ°(HCOOH)
ά = 46.1 / 404.2
ά = 0.114
Formula of dissociation constant:
K = (c ά2)/(1 – ά)
Plug the values we get
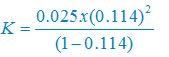
K = 3.67 × 10–4 mol per liter
Copyright @ ncerthelp.com A free educational website for CBSE, ICSE and UP board.